Researchers in Nuclear, Plasma, and Radiological Engineering, Bioengineering, and Chemistry at the University of Illinois have developed a sustainable way of forming carbon-carbon bonds, which is the foundation of all organic compounds. The researchers have demonstrated a novel metal-catalyst-free approach that could revolutionize organic chemistry. The process could lead to a new era in organic chemistry, as it eliminates the need for expensive, rare metals that are typically required as catalysts in bond-forming organic reactions.
The Research
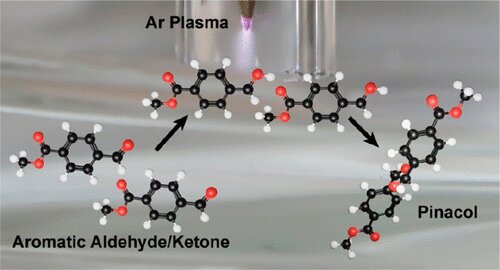
In the study, “Plasma Electrochemistry for Carbon−Carbon Bond Formation via Pinacol Coupling,” published in the Journal of the American Chemical Society, the team explains how they used a plasma-liquid process and electricity to generate solvated electrons to form carbon-carbon bonds in a pinacol coupling reaction. The formation of C-C bonds is extensively used in the production of many man-made chemicals such as pharmaceuticals and plastics.
According to the researchers, this is the first example of plasma-generated solvated electrons for an organic redox coupling reaction, and it offers a sustainable solution for similar reductive organic reactions. Typically, such reactions require metal reagents or catalysts that are not only scarce and costly, but also present safety or environmental issues and sometimes require heat in the reactive process.
The Process
The process produces electrons from argon gas and then injects these electrons into the solution to generate solvated electrons, a powerful chemical species typically generated by radiolysis, which requires complex equipment. The solvated electrons are generated with a DC power supply and a relatively simple electrolysis reactor that houses the electrodes and the solution where the organic substrates are. The process only requires electricity, and in the future, researchers hope to source the electricity from renewable sources like wind or solar or nuclear. The whole process is sustainable.
The Collaboration
The project is a result of an interdisciplinary collaboration between researchers in Nuclear, Plasma, and Radiological Engineering, Bioengineering, and Chemistry. Jian Wang, lead author of the study and a postdoctoral associate in the Moore group, brought his expertise in chemistry and materials science to the project and worked with plasma expert Scott Dubowsky, study co-author, and research scientist in the Sankaran group, to learn the plasma-liquid process and then identify an organic reaction to study.
Matthew Confer, another co-author and a postdoctoral researcher in the group of Rohit Bhargava, professor of bioengineering and faculty affiliate in chemistry, used his computational chemistry expertise to model how the pinacol product was formed from the solvated electron chemistry and radical reactions. The collaboration was essential for the success of the project.
Future Research
The researchers’ next step is to apply their process to another organic chemistry reaction and show that this approach is general and can be applied to different reactions. They also hope to find a reaction that is difficult to carry out, because it has a low yield, requires harsh conditions, or there is no active metal. The researchers hope to address one issue their study revealed – that yields can be limited by mass transfer limitations. The reaction occurs at the interface of a plasma and the solution, and for the substrate to reach the interface, it must diffuse. The researchers can address this issue by incorporating liquid flow, which will enhance mass transport by convection. Liquid flow will also potentially help them scale up the process, whereby they can make a product continuously.
The sustainable process for carbon-carbon bond formation that the researchers at the University of Illinois have developed, eliminates the need for expensive, rare metals that are typically required as catalysts in bond-forming organic reactions. This innovative process could lead to a new era in organic chemistry. It is the first example of plasma-generated solvated electrons for an organic redox coupling reaction and offers a sustainable solution for similar reductive organic reactions. The researchers’ next step is to apply their process to another organic chemistry reaction and show that this approach is general and can be applied to different reactions.


Leave a Reply