In the battle against cancer, scientists face the daunting task of identifying effective drug targets amidst the vast complexity of tumor cells. These cells rely on thousands of proteins to function, but only a fraction of them can be specifically targeted by drugs to treat cancer safely and efficiently. However, a groundbreaking study led by Scripps Research and the Broad Institute of Harvard and MIT has introduced a novel method to pinpoint potential drug targets that can have a broad impact on various types of cancer.
Published in Nature Chemical Biology on October 2, 2023, the research article details an innovative gene editing approach that alters over 13,000 potential drug targets to identify precisely which edits affect cell growth. By integrating this data with chemical proteomic information, the study identifies hundreds of potential drug targets, including ones that have never been explored before. The senior author of the study, Benjamin Cravatt, Ph.D., describes this approach as a groundbreaking strategy for discovering new cancer treatments and provides critical preclinical information on the protein sites that are most likely to impact cancer cell growth.
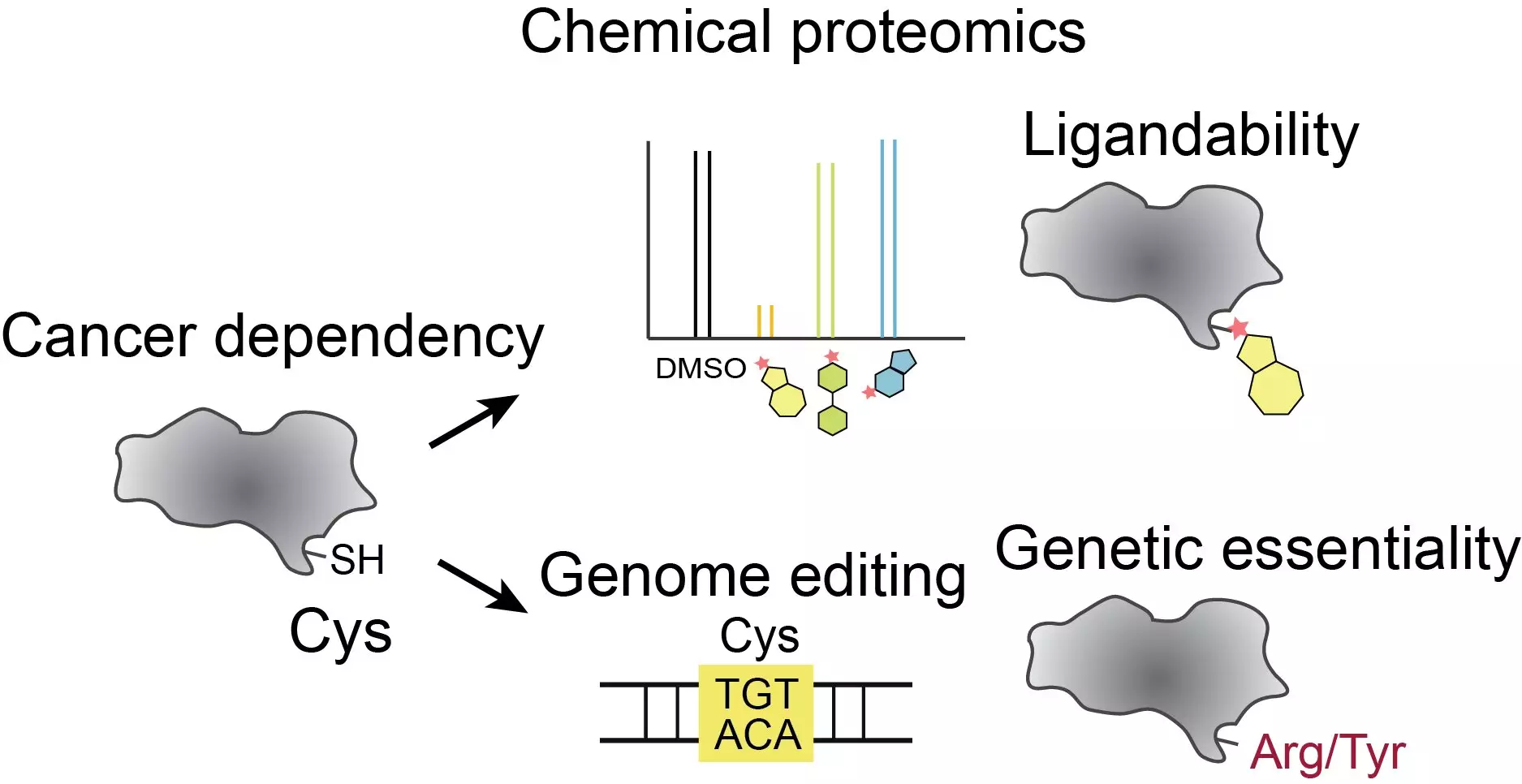
In recent years, chemists and pharmaceutical companies have focused on a class of drugs that permanently bind to cysteines—a type of amino acid crucial in human proteins. Cysteines have unique reactive chemistry that makes them an ideal target for drug development. However, with hundreds of thousands of cysteines scattered throughout human proteins, identifying the ones that can be targeted with drugs has proven to be an immense challenge. Even among the few thousand proteins recognized as vital to cancer cell growth, there are still over 13,000 cysteines to consider. Therefore, narrowing down the list of cysteines that significantly impact cancer-relevant proteins requires a more precise approach.
Inspired by Stuart Schreiber’s work on using human genetic variation to guide drug discovery, postdoctoral research associate Haoxin Li collaborated with David Liu at Scripps Research and the Broad Institute to combine precise genome engineering techniques with cutting-edge chemical proteomic tools. Li used base editing—a method for precisely modifying DNA letters—to introduce targeted amino acid changes in cancer cells. By creating mutations that affected a variety of cysteines, the team aimed to gain insights into which cysteines played the most critical role in cancer cells.
The research team edited 13,800 locations on more than 1,750 genes previously associated with cancer cell survival. In each case, they targeted a cysteine on the corresponding protein. The team then examined how cancer cells with these mutations proliferated. They also integrated their findings with new data on the “druggability” of these cysteines. Among the discoveries, they identified approximately 160 druggable cysteines that, when edited, influenced cancer cell growth. This highlights the potential for drugs targeting these cysteines to be effective in cancer treatment.
One of the most impactful edits involved a change to the cancer-dependency protein TOE1. Although TOE1 was previously known for its role in trimming the ends of RNA molecules, it had not been considered as a potential cancer drug target. However, Li and Cravatt’s team demonstrated that small molecules could be employed to target this previously overlooked vulnerability of cancer cells. This finding opens up exciting possibilities for the development of new compounds that can impede TOE1 and restrict cancer cell growth.
While further research is necessary to determine if drugs targeting TOE1 could be beneficial to human patients, the initial results showcase the effectiveness of editing cysteines in predicting potential drug targets. The research team intends to explore other novel targets revealed by their experiments and envisions the development of next-generation chemical genetic approaches to study druggable cysteines in diseases beyond cancer.
The study, titled “Assigning functionality to cysteine by base editing of cancer dependency genes,” was co-authored by researchers from Scripps Research, the Broad Institute of Harvard and MIT, Vividion Therapeutics, and UC San Diego. Jarrett Remsberg, Sang Joon Won, Kristen DeMeester, Evert Njomen, Daisuke Ogasawara, and Bruno Melillo contributed from Scripps Research. Kevin Zhao, Tony Huang, Stuart Schreiber, and David Liu represented the Broad Institute. Bingwen Lu and Gabriel Simon joined from Vividion Therapeutics. Finally, Tiantai Ma and Jens Lykke-Andersen contributed from UC San Diego.
This innovative study presents a promising avenue for identifying new drug targets for cancer treatment. By utilizing precision gene editing and integrating chemical proteomic data, the researchers have unlocked potential drug targets that were previously overlooked. While there is much work ahead to develop effective treatments based on these findings, this groundbreaking approach provides hope for the future of cancer therapeutics.


Leave a Reply