A groundbreaking technology has emerged to revolutionize the production of green hydrogen by addressing the limitations of current catalyst electrodes. Led by a team of researchers from the Department of Materials Science and Engineering and the School of Energy and Chemical Engineering at UNIST, along with collaborators from King Abdullah University of Science and Technology (KAUST), this new development has the potential to produce green hydrogen on a large scale at a relatively low cost.
Traditional methods of manufacturing electrochemical electrodes involved spraying a powder catalyst onto the electrode for fixation, which often led to issues such as uneven application, clumping, or detachment of the powder. However, the new carbon fabric electrocatalysts developed by the research team offer a more stable operation across large areas by utilizing a carbon fiber catalyst instead of a powder-type catalyst. This innovative design not only enhances stability but also boasts a lifespan 100 times longer than conventional electrodes.
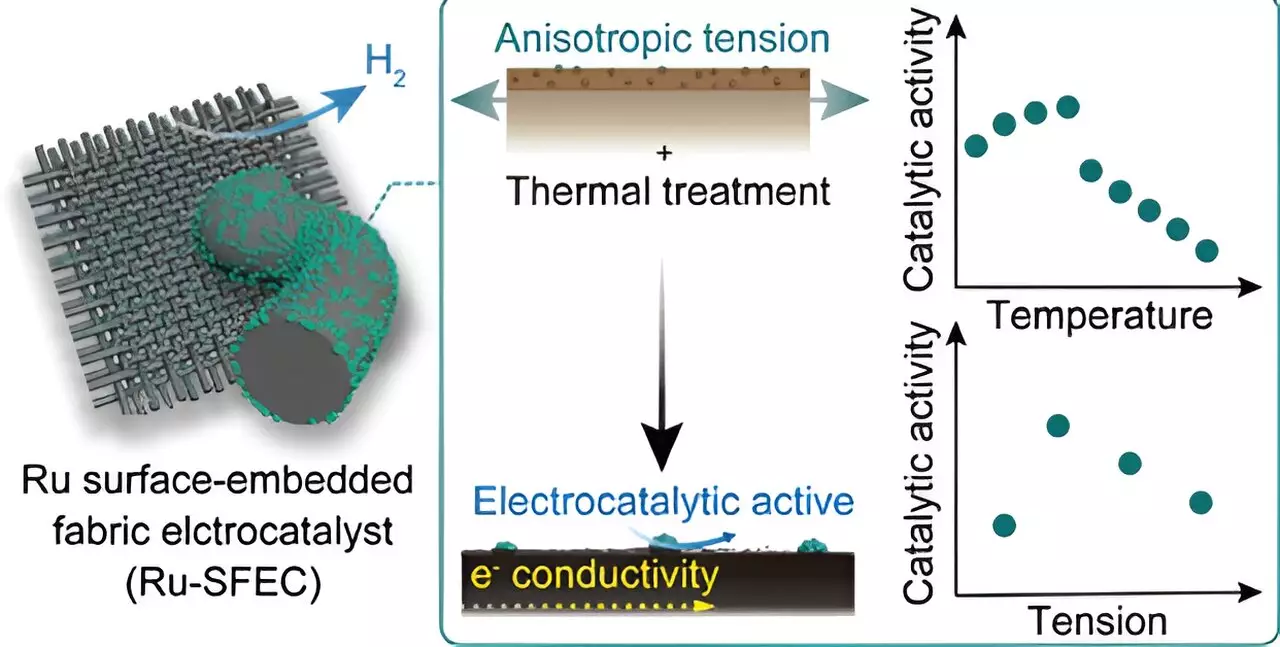
One of the key components of this advanced electrode design is the use of ruthenium instead of the more expensive platinum. By integrating ruthenium into the polymer precursor fiber during the manufacturing process, the researchers were able to enhance catalyst stability and reduce manufacturing costs significantly. The ruthenium surface-embedded fabric electrocatalysts (Ru-SFECs) demonstrated low overvoltage and energy consumption during the hydrogen generation process.
The carbon fiber-based electrochemical electrodes offer high thermal and electrical conductivity properties, as well as ease of use across large surfaces, making them a versatile material for future electrochemical reactions. By meticulously controlling catalyst metal separation and microcarbon structure, the researchers achieved maximum stability and efficiency, enabling the continuous production of catalyst fibers for direct industrial applications.
The development of stable, binder-free, and flexible electrocatalytic electrodes opens up possibilities for a wide range of catalytic reactions with different metals. Future research should focus on enhancing mechanical durability, electrical conductivity, and cost-effectiveness to further expand the applications of this technology. The continuous production of catalyst-supported carbon fibers on a semi-pilot line demonstrates the technological maturity suitable for real-world implementation.
The innovative approach taken by the research team in developing carbon fabric electrocatalysts embedded with highly functional catalysts represents a significant advancement in the field of green hydrogen production. By utilizing ruthenium instead of platinum and incorporating it into the polymer precursor fiber, they have created a cost-effective and efficient electrode design with remarkable stability and performance. This technology not only offers energy-efficient manufacturing processes but also reduces waste production, paving the way for a sustainable and eco-friendly future.


Leave a Reply