For over two centuries, scientists have grappled with the challenge of growing dolomite, a common mineral found in various geological formations. Dolomite is abundant in older rocks but remarkably absent in younger formations. However, a team of researchers from the University of Michigan and Hokkaido University in Japan has finally managed to grow dolomite in the laboratory. Their groundbreaking success has solved the long-standing geology mystery known as the “Dolomite Problem.” This achievement holds significant potential for understanding natural crystal growth and developing new strategies for manufacturing modern technological materials.
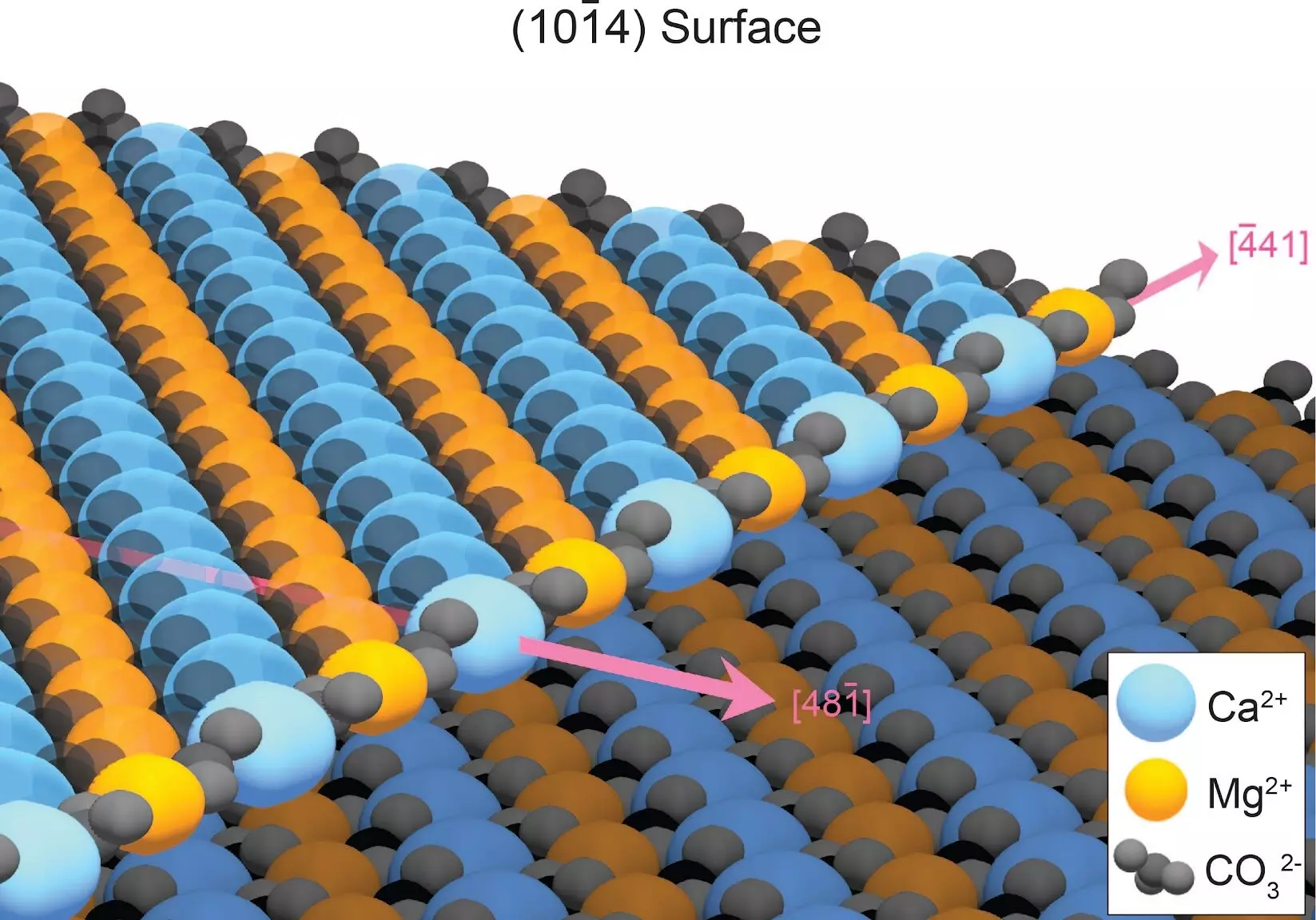
To understand why dolomite growth has been so elusive, it is essential to examine the mineral’s crystal structure. Unlike many other minerals, dolomite forms with alternating rows of calcium and magnesium atoms. When minerals grow in water, atoms usually deposit neatly along the growing crystal surface. However, calcium and magnesium atoms tend to attach randomly to the growing dolomite crystal, creating defects that hinder further dolomite layer formation. These defects slow down the growth process considerably, making it exceptionally time-consuming to develop ordered dolomite layers.
The researchers discovered that defects in the crystal structure were responsible for the slow growth of dolomite. Fortunately, these defects are not permanent but can be dissolved when the mineral is rinsed with water. By repeatedly rinsing away the defects, either through rain or tidal cycles, dolomite layers can form within a matter of years. This process, which occurs over geologic time, is responsible for the accumulation of dolomite mountains. The researchers successfully simulated dolomite growth by accurately calculating the strength of atomic attachments on an existing dolomite surface. This calculation involved analyzing the energy of every single interaction between electrons and atoms in the growing crystal.
Pioneering Advances in Simulating Dolomite Growth
Traditionally, such exhaustive calculations demanded significant computing power. However, the researchers made use of software developed at the University of Michigan’s Predictive Structure Materials Science (PRISMS) Center to expedite the process. This software enabled the researchers to predict the energies for different atomic arrangements by extrapolating from known arrangements. This shortcut significantly reduced the computing time required for simulating dolomite growth over geologic timescales, making the process more efficient and feasible.
To validate their theory, the research team collaborated with scientists from Hokkaido University. They utilized transmission electron microscopes and a unique feature of the electron beam that had the ability to split water and dissolve crystals. By subjecting a small dolomite crystal to the electron beam, the defects were gradually dissolved, allowing dolomite to grow. The experiment resulted in the growth of approximately 300 layers of dolomite, a significant breakthrough considering that no more than five layers had ever been grown in the lab before.
Implications for Technological Advancements
The resolution of the Dolomite Problem has far-reaching implications for various technological industries. Engineers can now apply the knowledge gained from this research to manufacture higher-quality materials for semiconductors, solar panels, batteries, and other tech devices. In the past, crystal growers attempted to eliminate defects by growing materials slowly, but with the newfound understanding of dolomite growth, engineers can develop alternative strategies to promote defect-free crystal growth in modern materials.
Concluding Thoughts
The successful growth of dolomite in the laboratory represents a significant scientific milestone. By overcoming the challenges of defect formation and slow growth, researchers have unlocked a wealth of knowledge that can be applied to various fields of study. With the insights gained from solving the Dolomite Problem, scientists can now explore new avenues for enhancing crystal growth techniques and manufacturing high-quality materials. This breakthrough holds promise not only for advancing our understanding of the natural world but also for catalyzing technological advancements that can shape the future.


Leave a Reply