Iridium oxide catalysts have been identified as being highly effective for water oxidation, making them a desirable option for green technologies. Researchers from SANKEN at Osaka University recently conducted a study to closely examine the workings of these catalysts in the Journal of the American Chemical Society.
During the study, the team utilized spectroscopy to uncover how the chemical species engaged in the iridium oxide-catalyzed oxygen evolution reaction (OER) interact with the surrounding solution. The OER is a crucial process in numerous clean energy applications, such as converting carbon dioxide into usable liquid fuels and producing green hydrogen through water electrolysis.
Catalytic processes can be intricate, involving a variety of intermediate species as they progress from the initial substance to the desired end product. By utilizing operando techniques, researchers were able to investigate these intermediates through spectroscopy while the reaction was occurring, providing valuable insights into the process.
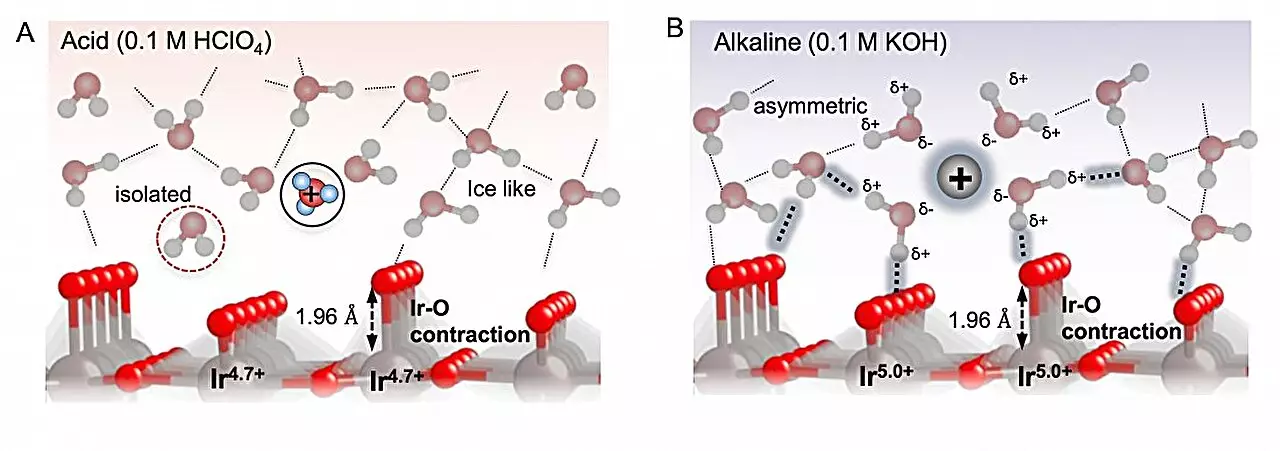
The team focused on an electrode with an iridium oxide surface to examine the oxidation of water molecules in solutions with varying pH levels. It was discovered that the interaction between the electrode surface and the oxygenated intermediates plays a critical role in the efficiency of the OER. By optimizing the catalyst material, researchers aimed to enhance the overall performance of the process.
The study revealed that the binding of reaction intermediates to the electrode is influenced by long-range interactions through the solution, a factor that is dependent on the pH level. In alkaline conditions, the proximity of water to the electrode impacted the interactions between the oxygenated species, affecting their binding to the surface. Despite the intermediates binding more strongly at higher pH levels, the interactions facilitated by interfacial water ultimately destabilized the oxygenated species, enabling the reaction to proceed.
Senior author Yu Katayama emphasized the importance of using operando spectroscopy and complementary techniques to gain a direct understanding of the species involved. This comprehensive approach broadens the understanding of catalyst performance beyond just electrode binding and is believed to be crucial in optimizing the kinetics of the OER.
The findings of the study are expected to contribute to enhancing the efficiency of water oxidation for green hydrogen production. By combining operando spectroscopy with complementary techniques, researchers may also gain valuable insights into the catalysis of various other processes, further advancing the field of green technologies.


Leave a Reply