A groundbreaking study conducted by RIKEN chemists has led to the isolation of an elusive structure involving two water molecules that had previously only been predicted but never observed. This significant finding has the potential to impact various fields, ranging from astrochemistry to the corrosion of metals. The research, which has been published in The Journal of Physical Chemistry Letters, sheds light on the ionization process of water and provides a deeper understanding of its implications in biological processes and radiation chemistry.
The Ionization of Water and its Significance
When a high-energy particle or photon collides with a water molecule, it can cause the ejection of an electron from the molecule, resulting in the formation of a positive ion (cation; H2O+) and a free electron. This ionization process has far-reaching consequences, serving as a trigger for a cascade of reactions with neighboring molecules. Understanding how water ionization proceeds is of paramount importance for physical chemists due to its pivotal role in biological processes and its impact on the corrosion of metals when water interfaces with them.
Theoretical Predictions and Experimental Validation
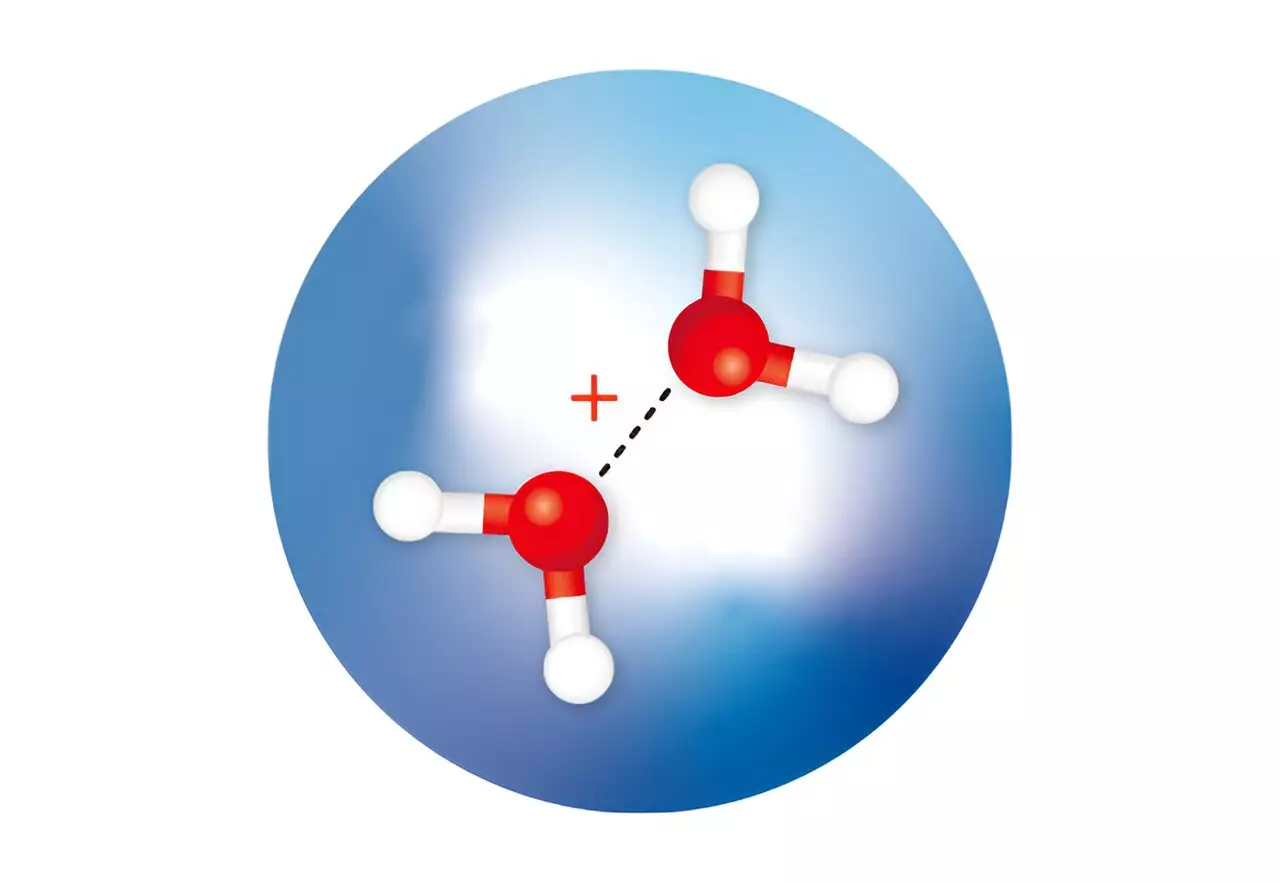
Theoretical calculations have long suggested that following the ionization of a water molecule, two isomers of a positively charged ion of a water dimer will rapidly form. A water dimer is formed when two water molecules are loosely connected by a weak bond. One isomer, known as H3O+·OH, has been observed and occurs when a proton is transferred from one water molecule to another. However, the other isomer, featuring a half-bond or hemibonded structure, namely (H2O·OH2)+, had remained elusive, with no confirmation through spectroscopic measurements.
RIKEN chemists, led by Susumu Kuma from the Atomic, Molecular and Optical Physics Laboratory, successfully isolated both water dimer ions by trapping them in minute droplets of cold helium. The researchers employed infrared spectroscopy to determine the structures of these ions. The ultracold environment was crucial for the isolation process, as the water molecules within the helium droplets rapidly cooled when the helium atoms evaporated from the droplet’s surface. This cooling process facilitated the stabilization of the metastable hemibonded isomer within the cold droplets.
Comparing Experimental Measurements to Theoretical Calculations
To validate their findings, Kuma and his team utilized computational and spectroscopic methods to probe the co-existence of the two isomers. The spectroscopic signatures of the water dimer ions closely resembled those of bare ions, without helium surrounding them. This indicates that direct comparisons can be made between measurements on the bare ions and results from quantum-chemical calculations, greatly assisting in the structural analysis of the dimers.
This groundbreaking discovery is expected to pave the way for further studies in the field of radiation chemistry. The isolation and characterization of the hemibonded water cations will enable researchers to delve deeper into primary events crucial for understanding the radiation chemistry of water. Expanding the size of the water complex cations in helium droplets is the next step for Kuma and his team, as they aim to uncover other yet-to-be-observed structures.
The isolation of the elusive water dimer ions represents a significant scientific achievement. The knowledge gained from this study has far-reaching implications, from advancing our understanding of astrochemistry to elucidating the corrosion processes occurring at interfaces between water and metals. This breakthrough serves as a reminder of the power of scientific discovery and the immense possibilities that lie ahead in unraveling the mysteries of the universe.


Leave a Reply