In the field of chemistry, the concept of chirality plays a significant role in both nature and laboratory synthesis. Chirality refers to the property exhibited by organic molecules that are either left- or right-handed, just like the human hands. This property is crucial in the development of drugs and enzymes, as the wrong “handedness” can render them ineffective. However, replicating the level of efficiency found in nature has always posed a challenge for chemists. Nonetheless, a recent study conducted at the University of California, Davis is making strides in bridging the gap between nature’s chemical efficiency and laboratory synthesis.
Chirality, as mentioned, refers to the property of molecules that share the same atomic makeup but are mirror images of each other. This property is akin to the distinction between left and right hands, as the mirror images cannot be superimposed on each other. In the realm of synthetic chemistry, the ability to create molecules with specific chiral forms is highly desirable, particularly in the development of drugs. Selective binding between chiral molecules and protein or enzyme targets is crucial for their desired pharmacological effects.
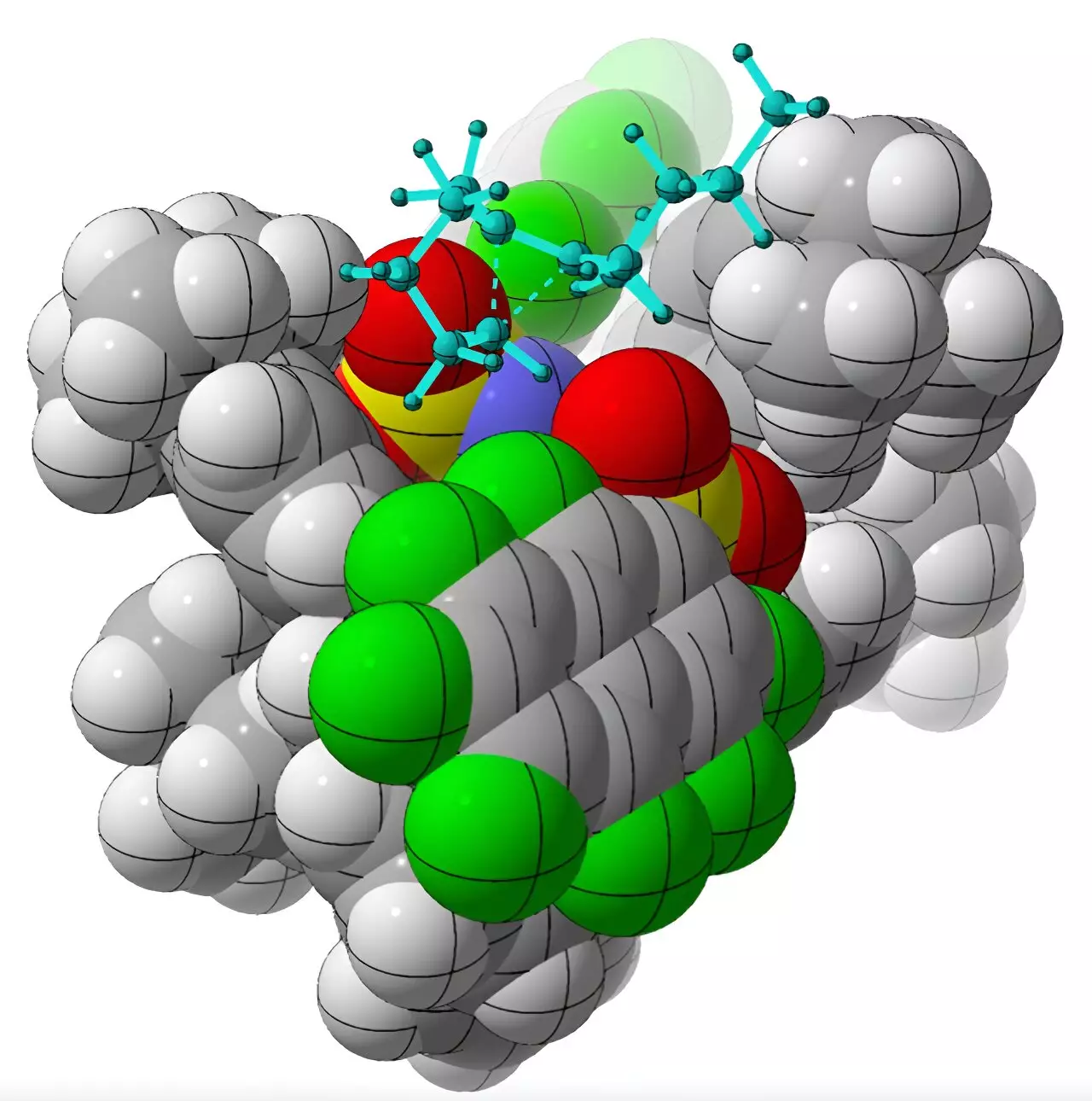
However, achieving selective chiral synthesis in a laboratory setting is no easy feat. Chiral molecules are often characterized by their “greasy-like” nature, with a positive charge smeared around them. This molecular property makes it challenging for chemical catalysts to bind to one chiral form over the other due to the lack of charged groups for the catalyst to interact with.
Nevertheless, researchers at the University of California, Davis have made a breakthrough in this area. By utilizing a chiral organic acid called imidodiphosphorimidate as a catalyst, the team successfully performed rearrangements of achiral alkenyl cycloalkanes. This process led to the production of specific chiral molecules known as cycloalkenes. Through computational methods, Professor Dean Tantillo and his colleagues were able to understand how the catalyst selectively produced one chiral form over the other.
This achievement is reminiscent of the behavior of enzymes involved in the production of hydrocarbon products called terpenes in nature. Professor Tantillo’s research focuses on mapping terpene reaction pathways using quantum mechanical methods. Understanding the stopping points and routes of carbocations during terpene formation is vital in comprehending and potentially manipulating these enzymes in the future.
The newly developed method holds promise for the production of both natural and nonnatural molecules. Petroleum serves as a vast source of hydrocarbons, and catalytically transforming these molecules into defined chiral forms would greatly enhance their value. However, the feasibility of such applications remains uncertain. Nevertheless, the advancements made in chiral chemistry offer a platform for further exploration and potential breakthroughs in the field.
The University of California, Davis’s research in chiral chemistry marks a significant milestone in the pursuit of mimicking nature’s efficiency. By successfully synthesizing specific chiral molecules using rearrangements of simple hydrocarbons and a novel chiral organic acid catalyst, the team has opened doors to a range of possibilities. Harnessing hydrocarbons for the production of medicines and materials could be revolutionized if the efficiency of natural processes can be replicated in the lab. As advancements in chiral chemistry continue to unfold, the potential implications for various industries become increasingly exciting.


Leave a Reply