Aspergillosis, caused by the fungal pathogen Aspergillus fumigatus, poses a significant threat to the lives of over 300,000 individuals annually. To address this medical concern, investigators at the University of Kansas have made groundbreaking advancements in deciphering a cluster of genes responsible for the production of sartorypyrones, a chemical associated with Aspergillosis. Their findings, published in Chemical Science, shed light on understanding the genes producing secondary metabolites in A. fumigatus and its fungal counterparts, potentially leading to the development of more effective antifungal drugs.
Aspergillosis is a severe pathogenic fungal infection primarily affecting immunocompromised individuals undergoing cancer treatment or residing in sub-Saharan Africa, where many people with AIDS lack access to medication. It is crucial to gain a better understanding of the secondary metabolites produced by A. fumigatus and similar fungi as they offer selective advantages, such as protection against factors like UV radiation and inhibition of competitive species. These metabolites also exhibit bioactivities that may have medicinal potential or contribute to the pathogenic effects, including immune system suppression.
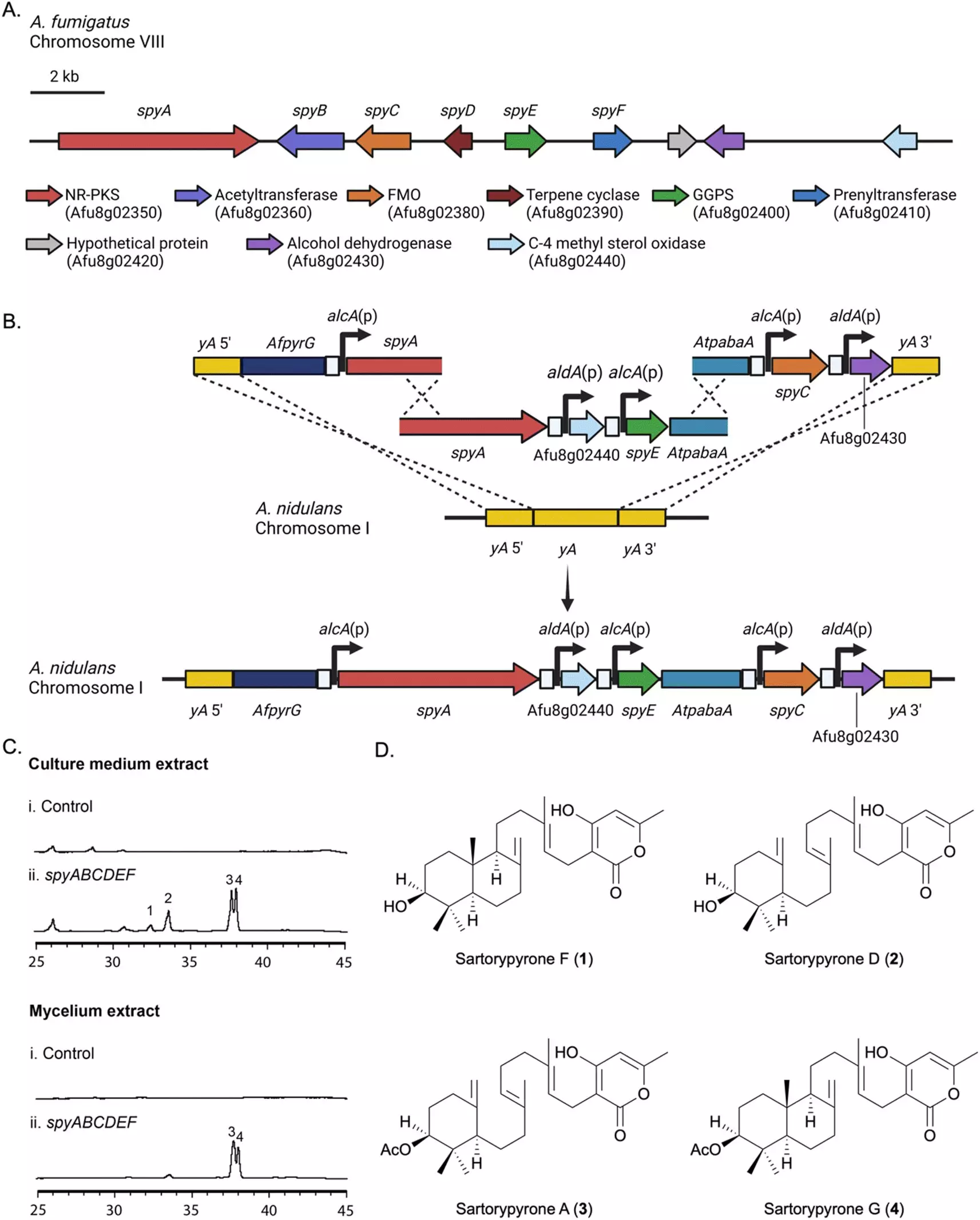
To analyze the genes responsible for secondary metabolite production in Aspergillus fumigatus, the researchers transferred a biosynthetic gene cluster (BGC) to a related strain of Aspergillus called A. nidulans. By activating these genes in A. nidulans, they were able to observe the compounds produced in the laboratory. This technique, known as “heterologous expression,” enabled the team to explore the synthesis of sartorypyrones, a group of compounds with limited prior knowledge of their production.
The researchers named the gene cluster responsible for sartorypyrones the “spy BGC” (short for sartorypyrones) and employed various analysis methods, including high-resolution electrospray ionization mass spectrometry, nuclear magnetic resonance, and microcrystal electron diffraction (MicroED), to identify 12 chemical products from the spy BGC. Fascinatingly, seven of these compounds had never been isolated before. The team proposed a biosynthetic pathway for this compound family, proving the efficacy of their gene cluster transfer approach.
The success of this study in deciphering the spy BGC may pave the way for further breakthroughs in understanding Aspergillus fumigatus and other fungal pathogens. The identification and understanding of additional secondary metabolite gene clusters could advance therapeutic interventions and provide insights into the mechanisms of infection. Moreover, the knowledge gained from this research may have implications for eco-friendly industrial applications. For example, the investigators’ previous work involving genetically modified A. nidulans demonstrated its potential to convert ocean plastics into raw materials for the pharmaceutical industry.
While the findings of this study offer promising possibilities for antifungal drug development, the economic realities of manufacturing such medications present a significant challenge. Berl Oakley, the corresponding author, acknowledges the pressing need for more antibiotics and antifungals. However, profitability factors heavily influence drug development, favoring compounds that can be given over an extended period rather than those with shorter treatment durations. Overcoming these barriers will require substantial financial incentives.
The University of Kansas investigators have made noteworthy strides in deciphering the genes responsible for sartorypyrone production in Aspergillus fumigatus. This research provides important insights into the biosynthetic pathways of secondary metabolites, shedding light on the potential development of more effective antifungal drugs. However, while these findings are significant, the economic challenges of manufacturing antifungal medications may hinder rapid drug development. Nonetheless, with continued research and collaborative efforts, the understanding of fungal pathogens and the development of therapies against Aspergillosis can progress, ultimately improving healthcare outcomes for those at risk.


Leave a Reply