Chemistry faces big challenges in controlling chemical reactions to create new products. This development has a significant impact on the industry, as it reduces waste in the manufacture of construction materials and improves the production of catalysts for speeding up chemical reactions. Polariton chemistry, which uses the tools of chemistry and quantum optics, has seen many experiments in optical cavities to manipulate the chemical reactivity of molecules at room temperature using electromagnetic fields. While some research teams have achieved success in modifying chemical reaction products in organic compounds, no team has yet come up with a general physical mechanism to describe the phenomenon and reproduce it for consistent measurements.
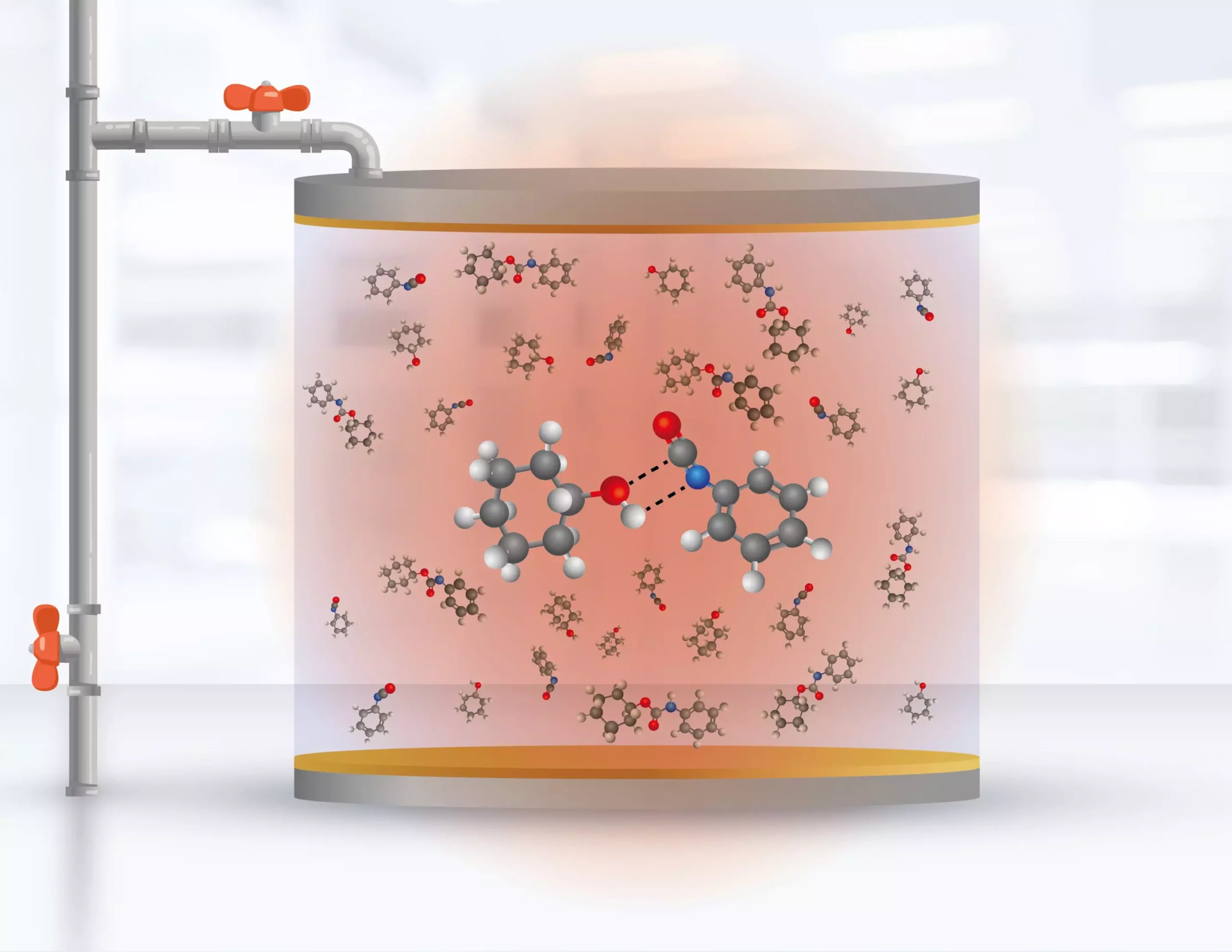
However, a team of researchers from the Universidad de Santiago in Chile and the laboratory of the chemistry division of the US Naval Research Laboratory have made a breakthrough in this field. Felipe Herrera and his team from the Millennium Institute for Research in Optics (MIRO) and researcher Blake Simpkins from the US Naval Research Laboratory report the manipulation of the formation rate of urethane molecules in a solution within an infrared cavity. The team’s discovery, published in the journal Science on June 16, 2023, provides theoretical and experimental proof that it is possible to selectively modify the reactivity of certain bonds in a chemical reaction at room temperature in a liquid solvent through the influence of the electromagnetic field vacuum in a narrow range of infrared frequencies.
Theoretical Understanding of Chemical Reactions
One of chemistry’s toughest challenges is controlling chemical reactions since the bonds that unite atoms in a molecule break and rearrange, forming new products. Energy is often required for this process, and physicochemical principles from the 19th century have helped us understand how energy transfer occurs according to the laws of thermodynamics. Principles of reactivity based on the structures of molecules, such as those proposed by Eyring, Evans, and Polanyi in 1935, are widely used in all areas of chemistry. These basic principles imply that each reaction between two molecules is independent of the other chemical reactions that may occur in a solution.
The electromagnetic vacuum creates correlations between the different chemical reactions that occur within the volume of the cavity. These correlations created by the electromagnetic field make the traditional assumptions of chemical reactivity questionable. The confirmation of the modification of the reaction rates through the interaction with the vacuum of the electromagnetic field confined inside the cavity is the experimental contribution of this study. The theoretical contribution is that by modifying the dynamics of the chemical bonds that mainly participate in the reaction through the infrared vacuum, it is possible to control the products.
The Team’s Research Process
Dr. Wonmi Ahn, a former postdoctoral fellow at the US Naval Research Laboratory who is now a professor at Bilkent University, conducted the first experiments in 2020. Blake Simpkins prepared new samples in 2021 to ensure the measurements were reproducible and improved the liquid cells where the chemical reactions occur. In the middle of that year, researcher Felipe Herrera began to have regular meetings with Simpkins to investigate possible theoretical answers to support the results obtained.
They decided to start from scratch and build a theory that takes all the physical aspects of quantum optics into consideration but reduces to the standard reactivity theory of theoretical chemistry under specific conditions. The result of the process is the publication “Modification of ground-state chemical reactivity via light-matter coherence in infrared cavities,” led by Simpkins (US Naval Research Laboratory) and Herrera (MIRO, Universidad de Santiago de Chile), with the participation of researcher Wonmi Ahn, researcher Johan Triana, and Ph.D. student Felipe Recabal, both part of the Molecular Quantum Technology group of MIRO, at USACH.
The team’s research opens new possibilities and scientific challenges. They need to develop a sufficiently simple and general theoretical and mathematical framework that any researcher in the world can use to interpret their experiments and design new types of measurements. Moving forward, Felipe Herrera reflects on his ambitions as a scientist moving across physics and chemistry. He aims to build a theory that unifies two of the most successful disciplines in modern science: chemical kinetics and quantum physics. Therefore, the team’s breakthrough discovery provides a promising direction for controlling chemical reactions through electromagnetic fields.


Leave a Reply