Researchers from the University of Edinburgh and the University of Cantabria have collaborated to develop an artificial intelligence (AI) model capable of identifying senolytic medicinal chemistry in familiar compounds. The scientists have published a paper titled “Discovery of senolytics using machine learning” in Nature Communications, detailing their efforts to search through over 4,300 scientifically described compounds to find the right chemical makeup that could help treat cellular senescence.
Cellular Senescence and its Importance
Cellular senescence refers to the process by which a cell stops multiplying, which can occur due to various reasons such as aging or damage. Senescence plays a crucial role in limiting tumor progression by not allowing damaged or mutated cells to proliferate. However, accumulating high levels of senescent cells with age is associated with various forms of disease, including osteoarthritis, lung disease, Alzheimer’s, dementia, and cancer.
AI Model to Identify Senolytic Medicinal Chemistry
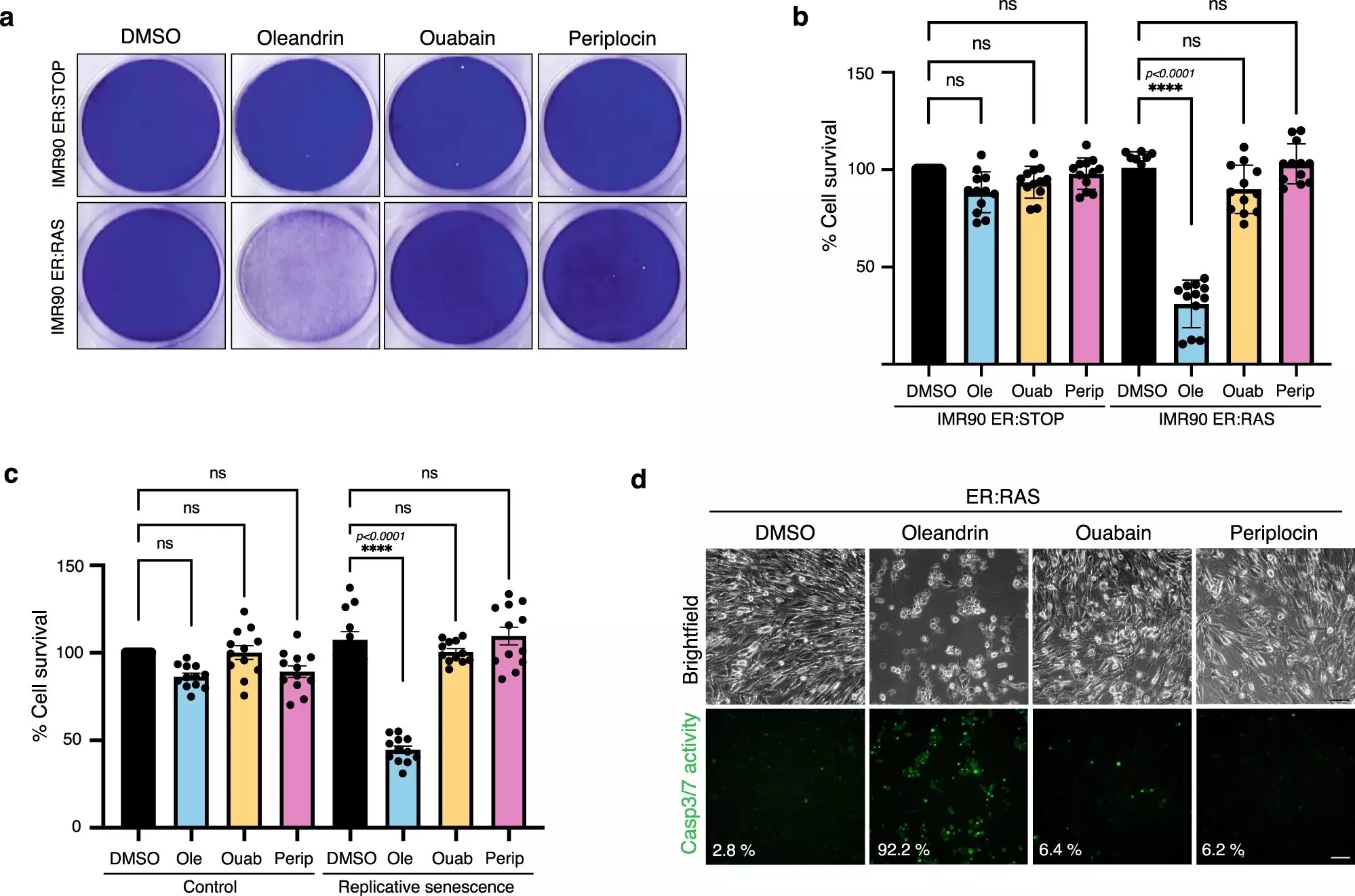
To identify senolytic medicinal compounds, the researchers screened 4,340 FDA-approved or clinical-stage compounds, using 58 previously identified senolytics and 2,465 compounds known to have no senolytic effects to train the AI model. The AI model identified three senolytic compounds, namely ginkgetin, periplocin, and oleandrin, all of which are present in herbal medicines. When tested on human cells, all three compounds were found to remove senescent cells without damaging healthy cells.
One of the three compounds, oleandrin, showed the most promising results. Oleandrin is a compound in the class of cardiac glycosides, which are used as heart medication to increase cardiac output during heart failure and in the treatment of hypotension and arrhythmias. The leading cardiac glycoside compound in use currently is ouabain, which is known to have relatively high toxicity and is a common cause of poisoning. In fact, ouabain is a crucial component of poison arrows used by Maasai warriors in Kenya.
Oleandrin showed a greater senolytic performance than ouabain, functioning at a low nanomolar range, inhibiting its canonical target, and activating its senolytic pathway with higher efficacy. Oleandrin did not affect the viability of normal cells at the tested concentrations, indicating promising senolytic potential. However, cardiotoxicity is still a concern with oleandrin. The researchers suggest that localized senolytic therapy, which involves using the compound directly on the site of damaged tissue, might make it an impactful treatment while reducing offsite risks.
Other Findings
In parallel, the researchers also showed that ex-vivo senolytics perfusion of human transplant livers preserves tissue architecture and regenerative capacity during cold storage.
The researchers’ AI model has identified potential senolytic medicinal compounds that could help treat cellular senescence. While oleandrin showed promising results, further research is necessary to determine its efficacy and safety. The study highlights the beneficial contribution AI can make in the hands of researchers and offers an exciting new compound to test in overcoming cellular senescence.


Leave a Reply