A groundbreaking discovery has been made by RIKEN chemists, who have successfully developed a highly active catalyst capable of synthesizing drug molecules within the body. This catalyst has shown promising results in mice, where it assembled an anticancer drug near tumors and effectively suppressed tumor growth. The potential implications of this development are immense, as it could revolutionize drug delivery systems and minimize the harmful side effects associated with conventional medicines.
In traditional drug delivery methods such as injections or pills, the active drug molecule circulates throughout the body, affecting both the target site and healthy tissues. Consequently, severe side effects may occur, leading to permanent damage and discontinuation of treatment. The ability to assemble drug molecules specifically at target sites within the body has the potential to enhance their efficacy while minimizing these detrimental side effects. Katsunori Tanaka, chief scientist of the RIKEN Biofunctional Synthetic Chemistry Laboratory, highlights the importance of a biocompatible biocatalysis system that can perform drug synthesis near target sites, as it would enable the treatment of diseases without harming healthy tissues.
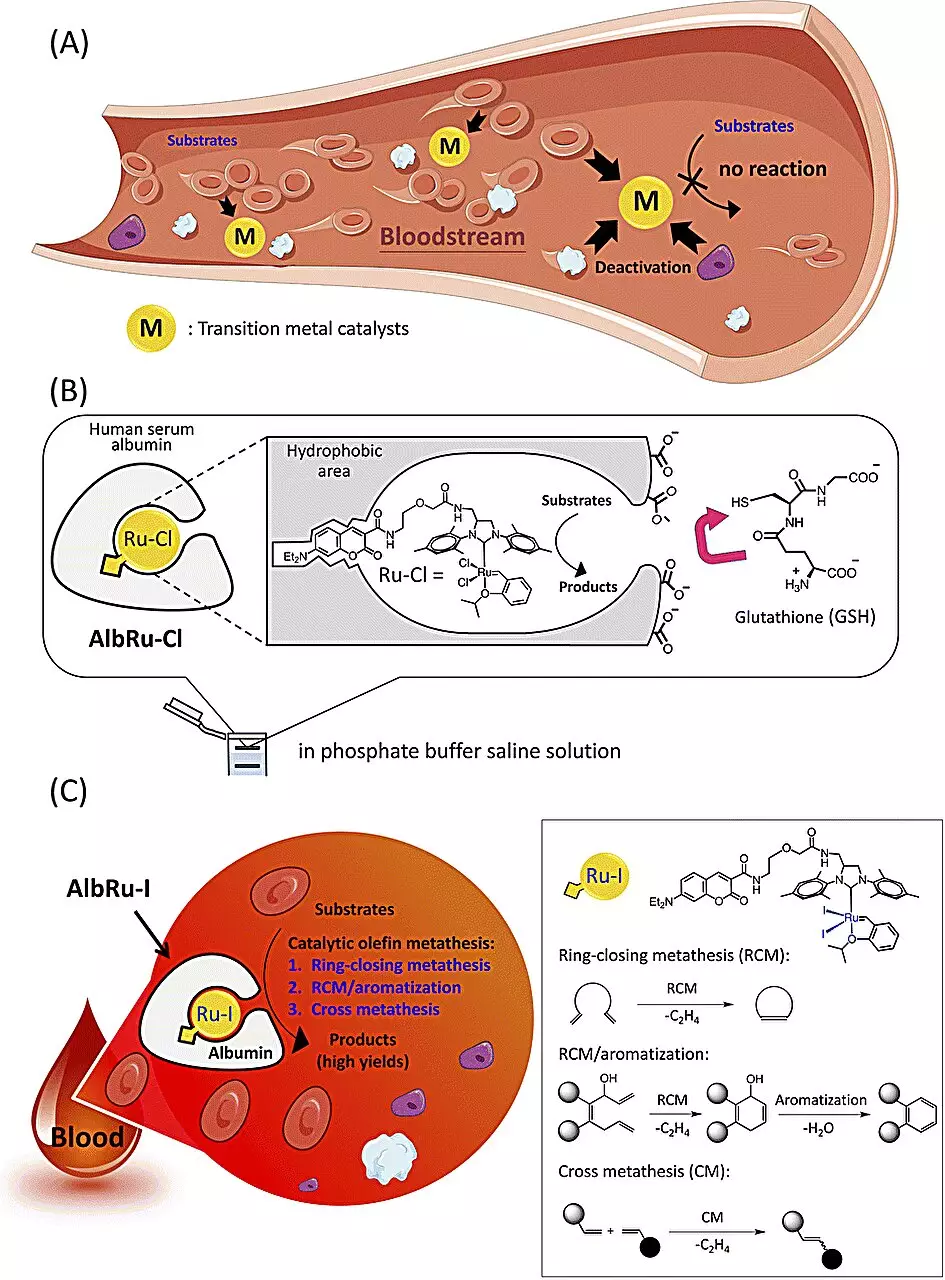
The RIKEN team focused on utilizing olefin metathesis, a catalytic chemical reaction known for efficiently constructing carbon-carbon double bonds in drug synthesis. Tanaka explains the potential of olefin metathesis in the body, as it could facilitate the synthesis of various types of drugs. However, most chemical catalysts are deactivated by biomolecules in the bloodstream, posing a significant challenge. To overcome this obstacle, the team encapsulated a ruthenium-based olefin metathesis catalyst in a protective protein called human serum albumin.
The researchers demonstrated that a ruthenium iodide complex, known as AlbRuI, within human serum albumin resulted in a highly efficient artificial metalloenzyme. At low catalyst concentrations, AlbRuI catalyzed multiple types of olefin metathesis reactions in blood with high yields. Furthermore, AlbRuI exhibited remarkable stability for 24 hours in blood, expanding the biocompatibility of artificial metalloenzymes and providing new opportunities for catalytic reactions in blood. This breakthrough paves the way for the development of general, metal-based artificial metalloenzymes for various catalytic reactions.
In addition to its catalytic properties, AlbRuI demonstrated its potential in tumor treatment. A low dose of cancer-targeting AlbRuI significantly inhibited tumor growth in mice by synthesizing an antitumor drug directly near the tumor site. This localized drug assembly provides higher efficacy in tumor suppression, further highlighting the versatility and potential applications of AlbRuI.
The RIKEN team has ambitious plans for the catalyst’s future applications. They aim to utilize AlbRuI for the synthesis of diverse bioactive molecules, potentially opening doors to more advanced and effective treatments. This breakthrough, if successfully translated into clinical practice, could revolutionize the field of medicine by providing targeted drug assembly and minimizing the harmful side effects associated with conventional drug delivery methods. However, further research and development are necessary to ensure the safety and efficacy of this breakthrough technology.
The development of a highly active catalyst capable of synthesizing drug molecules within the body is an exceptional breakthrough in the field of medicine. The potential to assemble drugs specifically at target sites in the body holds promise for enhancing their effectiveness while minimizing side effects. The AlbRuI catalyst, encapsulated within human serum albumin, has demonstrated remarkable stability and catalytic efficiency, making it a game-changer in drug synthesis. Furthermore, its ability to inhibit tumor growth through localized drug assembly showcases its potential applications in cancer treatment. The future of this catalyst looks promising, with the possibility of synthesizing various bioactive molecules, leading to more advanced and targeted therapies. As research progresses, we eagerly await the next wave of innovations in the field of drug synthesis and delivery.


Leave a Reply