Water contamination by nitrates poses a dire threat not only to ecosystems but also to public health. High levels of nitrates have been linked to various health issues, including methemoglobinemia, or “blue baby syndrome,” which affects infants. The sources of nitrate pollution often stem from agricultural runoff, sewage effluents, and industrial discharges, rendering clean water sources increasingly scarce. As society grapples with the repercussions of this contamination, the need for innovative filtration technologies becomes ever more vital.
In response to this troubling scenario, a team at Yale University has made significant headway. Through the pioneering work of Lea Winter, an assistant professor of chemical and environmental engineering, a new mechanism for effectively eliminating nitrates from drinking water has been developed. This novel method promises not only to address the immediate issue of nitrate removal but also to significantly improve the efficiency of existing techniques.
Existing Solutions and Their Limitations
Historically, there have been primarily two approaches to managing nitrates in water: separating them from the liquid or destroying them outright. Traditional separation leaves behind a concentrated waste product, which often returns to the environment, perpetuating the cycle of pollution. On the other hand, known destruction methods include biological denitrification processes primarily performed in wastewater treatment facilities. While effective, these biological processes face significant challenges due to their inherent sensitivity to environmental conditions—any small alteration in temperature, chemical composition, or pH can destabilize the strategy, slowing down or halting the nitrate removal process entirely.
Electrocatalytic approaches have emerged as an alternative. These methods involve applying electrical currents to facilitate the breakdown of nitrates. However, the conventional structures used in electrochemical systems often suffer from inefficiencies related to fluid movement, impeding the rapid conversion of nitrates. As research progresses, it’s clear that despite their efficacy, previous methods require considerable advancements to meet contemporary water quality demands.
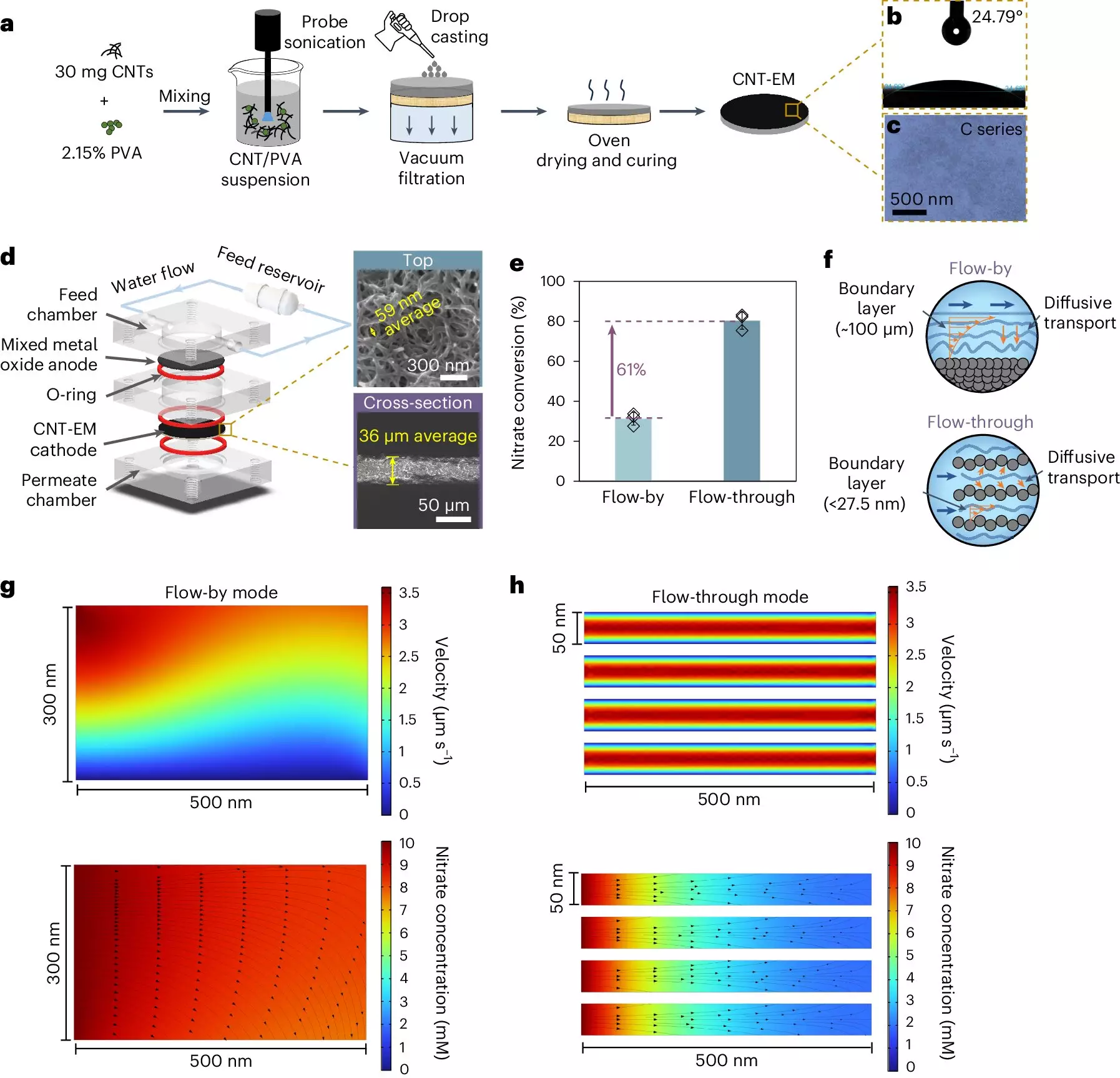
The Breakthrough: Electrified Membranes Made from Carbon Nanotubes
Winter’s innovative solution relies on electrified membranes composed of carbon nanotubes, a type of nanomaterial known for its strong electrical and mechanical properties. This new configuration offers superior performance compared to standard two-dimensional electrochemical systems that utilize flat plate electrodes. Winter highlights a fundamental flaw in conventional systems: the boundary layer that forms near the electrode surface, typically extending about 100 micrometers thick, slows down the movement and reaction of nitrates.
In contrast, the membranes created in Winter’s lab boast pores measuring just 50 nanometers—approximately 2,000 times smaller than those found in traditional setups. This dramatic reduction in pore size effectively diminishes the boundary layer thickness, allowing nitrates to reach the electrode surface much more rapidly. As a result, higher rates of conversion can be achieved without the need for metal catalysts, usually the cornerstone of such electrochemical processes.
Speeding up the Cleansing Process
One of the most striking aspects of Winter’s new approach is the speed at which nitrate conversion can be achieved. Conventional methods often require hours, sometimes even days, to reach 80-90% reduction of nitrates. Winter’s team, however, reported achieving similar results within a mere 15 seconds. This unprecedented efficiency illustrates a significant leap toward rapid and effective water purification, which is particularly crucial in times of urgent need. Imagine if municipalities faced with nitrate-laden water supplies could quickly provide safe drinking water to their communities!
This breakthrough is not just a theoretical success; in a real-world application, Winter’s team tested the system using water samples from Lake Wintergreen, aiming to ascertain its effectiveness with lower concentrations representative of naturally polluted waters. The ability to efficiently treat lower concentrations further enhances the technology’s relevance to real-world scenarios, ensuring that it is a feasible solution to nitrate contamination across various contexts.
A Forward-Looking Approach
The strides made by Yale researchers offer a glimpse of hope for tackling nitrate pollution that plagues water systems globally. As this technology continues to evolve, it may play a crucial role in enhancing global water security. While challenges will persist in the journey toward universal application, especially in scaling up production and deployment, Winter’s electrified membranes represent an inspiring challenge to the status quo of water purification strategies. By focusing on innovative materials and processes, we may significantly improve the pathways to clean drinking water, directly impacting public health and environmental sustainability. This promising technology could chart new directions in the fight against water pollution and the creation of safe drinking water for all.

Leave a Reply