
In a ground-breaking advancement in organic chemistry, Professor Max Martin Hansmann and his research team have made a significant breakthrough with a newly synthesized reagent designed for the selective addition of carbon atoms to molecular structures. Such innovations not only hold promise for immediate applications in synthetic chemistry but also pave the way for more complex molecular architectures in pharmaceuticals. Published in the renowned journal *Science*, this research is fundamentally reshaping our understanding of carbon transfer processes.
This initiative stems from a broader effort supported by the European Research Council (ERC) Starting Grant awarded to Professor Hansmann in 2022, underscoring the role of institutional funding in fostering cutting-edge scientific exploration. Dr. Taichi Koike, the postdoctoral researcher leading this commendable work as the first author, exemplifies how interdisciplinary collaboration can energize research dynamism and drive substantive developments in organic chemistry.
Professor Hansmann emphatically states that the precise modification of molecules on a single-atom level represents one of the most refined transformations achievable in organic chemistry. This statement encapsulates the pioneering spirit that defines his research team’s work. Their newly developed reagent is not merely a tool for carbon atom transfer; it symbolizes a leap in the chemistry landscape, offering researchers access to complex pharmaceutical compounds with substantially shorter and safer synthetic sequences.
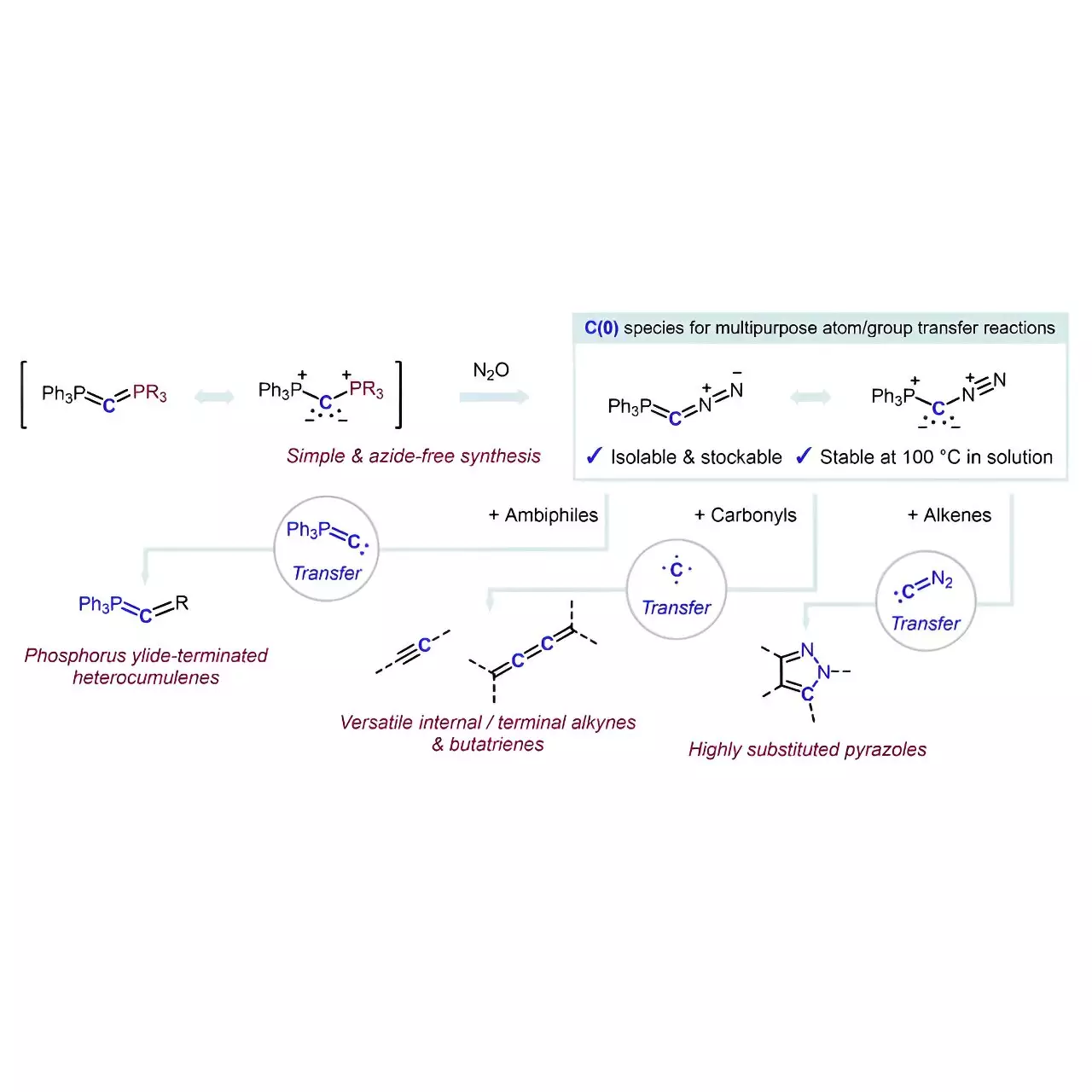
Despite the potential promise of introducing carbon atoms with precision, selective reagent development presents significant challenges. Typically, carbon sources in organic synthesis involve cumbersome procedures that can introduce safety risks. However, Hansmann’s team has ingeniously synthesized a reagent that serves dual purposes, acting both as a carbon source and a multipurpose transfer agent. The underlying innovation lies in the stabilization technique of the carbon atom itself, which involves coordinating it with two neutral, electron-donating groups, paving the way for unprecedented reactivity.
The significance of the reagent’s mechanism is startlingly elegant. What remains intriguing is the creation of carbones (L1→C←L2) that, until this research, had not been extensively utilized as carbon sources. The innovative reagent, which is flanked by simple and labile groups (PPh3 and N2), allows for a high level of reactivity without the complications that often accompany traditional diazo compounds. Hansmann’s approach sidesteps the usual requirement for azides, which are known hazardous materials in many laboratory environments.
The resultant compound, Ph3P=C=N2, synthesized through a formal PPh3/N2 exchange reaction, exemplifies simplicity and efficacy in its synthesis. This creativity not only enhances the safe production of the reagent but also facilitates a diverse range of applications. The study describes how the reagent can selectively transfer carbon fragments, leading to transformative reactions that yield phosphorus ylide-terminated heterocumulenes and enable sophisticated alkenes to undergo functionalization into multi-substituted pyrazoles.
Professor Hansmann’s research extends beyond immediate applications; it ventures into unexplored territories in carbon chemistry. The implications of this reagent can significantly streamline the late-stage functionalization of complex molecules. Such advancements mean that the chemical community could efficiently construct complicated pharmaceuticals, which are often hindered by tedious multi-step synthetic routes today.
Furthermore, the confidence expressed by Professor Hansmann regarding exploring the reactivity of this novel reagent indicates a vibrant future for carbon transfer methodologies within the domain of organic chemistry. The implications of this breakthrough are poised to resonate throughout the scientific community, encouraging further research and exploration into previously challenging pathways in synthetic organic chemistry.
Continuous innovation alongside scientific rigor positions Hansmann’s work at the forefront of a transformative era in chemistry. As we navigate the complexities of molecular synthesis, initiatives like this promise to unravel new dimensions of chemical understanding and application, marking a bright horizon for future explorations.
In the battle against climate change, reforestation has emerged as a beacon of hope, particularly…
Mars continuously captivates scientists and enthusiasts alike with its astonishing geological phenomena. Recently, the Perseverance…
In the rapidly advancing realm of technology, the emergence of quantum computing heralds a transformative…
Opioids have long been heralded as among the most effective solutions for alleviating intense pain,…
As of late March 2025, the measles outbreak that originated in west Texas has spiraled…
Water contamination by nitrates poses a dire threat not only to ecosystems but also to…
This website uses cookies.